Co element ions1/18/2024 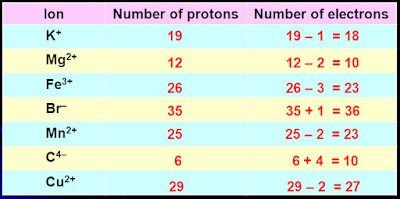
… In contrast, the secondary coordination sphere normally utilizes non-covalent interactions and is associated with determining chemical selectivity. The primary coordination sphere is dominated by covalent interactions between donor atoms on ligands and metal ions. What is primary and secondary coordination sphere? … Cobalt(2+) PubChem CIDĬobalt(2+) Cobaltous cation cobalt ion Co2+ Cobaltous ion More… 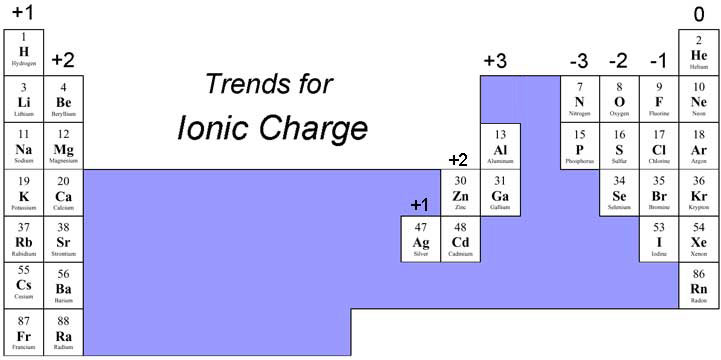
What is the formula for cobalt?Ĭo + 2 Please visit the Cobalt element page for information specific to the chemical element of the periodic table. Although long considered an insignificant and potentially toxic waste product of heme catabolism, CO is now recognized as a key signaling molecule that regulates numerous cardiovascular functions. What is the function of carbon monoxide?Ĭarbon monoxide (CO) is an endogenously derived gas formed from the breakdown of heme by the enzyme heme oxygenase. They form when a positively charged metal ion comes into contact with the oxygen atoms of the carbonate ion. Read More: What does it mean to be an involved citizen? How is carbonate formed?Ĭarbonates are the salts of carbonic acids. What is meant by the coordination number?Ĭoordination number, also called Ligancy, the number of atoms, ions, or molecules that a central atom or ion holds as its nearest neighbours in a complex or coordination compound or in a crystal. (physical chemistry) An ion, such as found in an ionized gas, whose properties, such as spectrum and magnetic moment, are not significantly affected by other atoms, ions, or molecules nearby. Occasionally, ligands can be cations (e.g., NO +, N 2H 5 +) and electron-pair acceptors. What can act as a ligand?Įxamples of common ligands are the neutral molecules water (H 2O), ammonia (NH 3), and carbon monoxide (CO) and the anions cyanide (CN –), chloride (Cl –), and hydroxide (OH –). Molecules in the sample that are negatively charged will “stick and other molecules will pass through quickly. … On the other hand, in anion exchange chromatography, the chemical groups attached to the beads are positively charged and the counter-ions are negatively charged. What is the role of counter-ions in ion exchange chromatography?Įach charged molecule has a counter-ion. In some cases, especially in the assembly of silver(I) coordination polymers, the counter-ion can play a key role in the formation of final supramolecular structure. In a general way, counter-ions can affect the crystallization of coordination polymer products, leading to different coordination structures. … Addition of a divalent ion leads to the formation of a mixed counterion layer and a decrease in the concentration of monovalent ions at the micelle/ solution surface (6,7). 
The valency of a counterion plays, for electrostatic reasons, an important role in the binding process. Read More: What are examples of conventional energy? Number How do you find the charge of CO? What is the charge for CA? 
What is the element of co?Ĭobalt cobalt (Co), chemical element, ferromagnetic metal of Group 9 (VIIIb) of the periodic table, used especially for heat-resistant and magnetic alloys. The second coordination sphere consists of molecules and ions that attached in various ways to the first coordination sphere. In coordination chemistry, the first coordination sphere refers to the array of molecules and ions (the ligands) directly attached to the central metal atom. What is a co ion?Īny ion of low molecular mass present in a colloidal solution or substance that has a charge of the same sign as the (net) charge of a (specified) colloidal ion under particular conditions. In certain colloids, the charge on the surface of colloidal particles is neutralized by oppositely charged counter ions in the surrounding solution. For example, in a crystal of sodium chloride, the chloride ions can be regarded as counter ions to the sodium ions. What is counter ion chemistry?Īn ion of opposite charge to a given ion. … Thus, the counterion to an anion will be a cation, and vice versa. counter ion, and sometimes written as two words) is the ion that accompanies an ionic species in order to maintain electric neutrality. What is meant by counter ions?Ī counterion (pronounced as two words, i.e. ZERO The formal charge of the carbon monoxide molecule is ZERO. As nouns the difference between coion and counterion is that coion is (chemistry) any ion of the same charge as another in a solution or other electrochemical system while counterion is (chemistry) any ion of opposite charge to another in a solution or other electrochemical system.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
